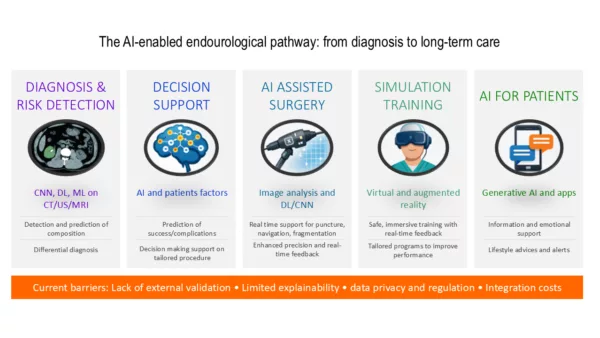
AI in endourology Is it ready for clinical application?
Artificial intelligence (AI) is no longer a futuristic concept in endourology, as it is already influencing diagnosis, operative planning, surgical training, and patient engagement. With numerous research and technological innovations in both urolithiasis and BPH management, discussion on the role and applicability of AI is gaining a central position in the endourological roundtable. However, its real-life applicability and routine clinical implementation still depend on several factors, including robust external validation, explainability, and seamless integration into clinical workfl ows.
From detection to decision-making
AI has demonstrated outstanding performance in stone disease diagnostics. A recent systematic review including 71 original studies reported a mean precision of 96.9 % for stone detection using CT, KUB and ultrasound imaging, with stone-composition prediction reaching 88.2 % accuracy, consistently outperforming conventional radiological assessment. These tools have the potential to standardise reporting, reduce inter-reader variability, and improve patient selection for SWL, URS and PCNL. On paper, deep learning (DL) and machine learning (ML) have the potential to reduce the workflow while improving timing and accuracy of stone detection, differential diagnosis and stone composition. Moreover, with integration of patient-specific characteristics, AI tools could be able to foresee the probability of spontaneous stone passage, stone recurrence, as well as help the decision-making process towards the choice of a tailored procedure.
Yet, its translation into daily practice remains in question. The EAU/YAU review on AI for shockwave lithotripsy identifi ed 17 studies, most reporting AUCs >0.9 for stone-free rate prediction. However, wide heterogeneity in model inputs and the predominance of single-centre datasets limit generalisability and highlight the urgent need for multicentre validation and standardised reporting frameworks. With such limitations, many applications remain theoretical rather than clinically deployable. Nevertheless, with increasing international collaboration and prospective data collection, these predictive tools may achieve adequate standardisation and reproducibility.
Similarly, the integration of DL on prostatic tissue MRI has been proved helpful in discriminating between benign hyperplastic tissue suspicious nodules requiring further investigation. Recent studies have in fact improved diagnostic accuracy by integrating imaging features with pathology data. Despite this, questions remain regarding the real-world reliability, reproducibility, and the extent to which clinicians should rely on algorithmic decision-making in the absence of defi nitive pathological confirmation
Predicting complications: learning from big data
AI is beginning to redefi ne peri-operative risk stratification. The FLEXOR registry analysis trained 15 ML algorithms on over 6,500 ureteroscopy cases, achieving up to 99.1 % accuracy for postoperative sepsis prediction and >95 % accuracy for bleeding and collecting-system injury. Crucially, explainable AI identified clinically intuitive predictors such as stone size, residual fragments, and pre-operative urine culture, enhancing clinician trust in model outputs. These results are currently being translated into digital predictive tools accessible across devices.
Further prospective validation will determine their real-world utility. Importantly, ML-based tools do not replace existing urological nomograms but rather expand on them by analysing vast datasets and capturing complex inter-relationships beyond traditional linear statistics. As such, ML-based nomograms may ultimately surpass classical predictive models in reliability and clinical relevance. Similarly, ML has been applied also for prediction of BPH surgical outcomes and complications. An EAU-Endourology collaborative study involving 3828 patients developed ML models to predict incontinence following endoscopic enucleation of the prostate. With promising results, authors achieved sensitivity exceeding 95%, allowing surgeons to identify high-risk individuals preoperatively and personalise follow-up pathways. These tools could assist clinicians not only in the decision-making process, but also during informed consent by providing personalised risk profi les tailored to individual patient characteristics.
In the operating theatre and the training lab
The EAU Section of Endourology has highlighted the emerging role of AI in endoscopic stone surgery, including automated calyceal puncture guidance, stone recognition, and real-time fragmentation feedback. These systems promise to enhance safety, precision, and reproducibility across centres. Equally transformative is AI-enabled training. A 2025 World Journal of Urology review demonstrated that virtual-reality (VR) simulators signifi cantly improve procedural performance in BPH surgery, with high fi delity and safety ratings. VR is therefore likely to become a core component of endourological education rather than a supplementary tool.
Empowering patients through AI
AI is also reshaping the patient experience. The recent evaluation of the UroGPT™ chatbot showed that over 70 % of kidney stone formers strongly agreed it improved their understanding, with a Net Promoter Score exceeding +75, comparable to leading consumer digital platforms. The use of generative AI platforms is spreading worldwide and people are increasingly turning to intelligent platforms for more information and support. On one hand this can bring concerns on the quality of information provided, and the possible negative role on patients’ expectations and disinformation, but on the other hand it could be a major opportunity to improve patients’ involvement in their own therapeutic process.
The development of regulated, controlled and certified generative AI platforms, such as UroGPT, represent in fact a drastic shift toward a professional consultation that is reliable, always available and affordable. Providing simple but secure information and support to patients through similar channels could alleviate the pressure on healthcare systems. It may also enhance service quality and improve the detection of patients and cases with red fl ags who need further medical advice. This kind of innovation and application will eventually show the growing role of AI in shared decision-making, lifestyle coaching, and recurrence prevention.
Barriers to routine implementation
Despite impressive technical performance, major challenges persist. There is a need for external validation and prospective trials, that actually prove the reliability and replicability of fi ndings. Current literature evidence is in fact limited by the retrospective nature, heterogeneity biases and missing data from pre-existing databases. There is also a lack of explainability in many of the proposed models, that raises concerns on the AI algorithms and development process, thus reducing clinician trust. Ethical and legal concerns regarding data privacy, bias, and accountability remain unresolved. One of the main challenges for clinical application of AI relies on regulation and standardisation. A recent European Urology Focus review on BPH underscores that without standardised reporting guidelines and multicentre validation, widespread adoption will remain aspirational. Finally, there are signifi cant costs that come with technological improvement, as well as need for available infrastructures and fi nancial sources. Without these, AI integration in pre-existing systems will be severely limited, particularly for low-resource settings.
Outlook: adjunct today, cornerstone tomorrow
So, is AI ready for clinical application in endourology?
- Yes, as a powerful adjunct.
- But not yet, as a universal standard of care.
AI already matches or surpasses expert performance in many diagnostic and pre-operative processes such as stone detection, complication prediction, as well as in surgical training. In data-rich, high-volume centres it is already improving care. However, only when explainable, prospectively validated, and seamlessly integrated into workfl ows will AI become a core, trusted component of endourological practice.
The next phase belongs to collaborative multicentre trials, EAU-endorsed standards, and clinician-driven development. If these standards are reached, AI will evolve not as a disruptive force, but as an enabling partner in precision endourology.
This was first printed in the EAU26 Congress News.