Is there a second, digital, me? PIONEERing big data and systems medicine technologies in prostate cancer research and care
By Dr. Charles Auffray (Vourles, FR)
This article reflects the highlights of the lecture Dr. Auffray gave at the EAU20 Virtual Congress on Sunday 19 July. His presentation can be found in the EAU20 Resource Centre.
Over the past two decades, advances in engineering and computer sciences have provided increasingly efficient and affordable access to high-dimensional functional genomics data as global measures of metabolomic, proteomic, transcriptomic, genomic, genetic, and epigenetic features. Their integration with biological, clinical, environmental, and lifestyle assessments using iterative statistical analyses, computational modelling, and experimental validation has enabled systems biology approaches, which started to transform biomedical research and clinical practice. This is triggering the transition from a reactive to a proactive practice of medicine, revolutionising how medicine is practised in the 21st century.1-10
The effective development of participatory, personalised, predictive, and preventive (P4) systems medicine was made possible by harmonisation of experimental and computational methods for data, information and knowledge collection, storage, analysis, integration, and sharing on a big data scale. High-performance cloud computing infrastructures were used11, supporting ongoing community efforts of biologists, medical doctors, and computer scientists for the development of systems medicine disease maps using an advanced intelligence approach for multi-level representation of domain expert knowledge and experimental data in a human and machine readable format (figure 1).12-13
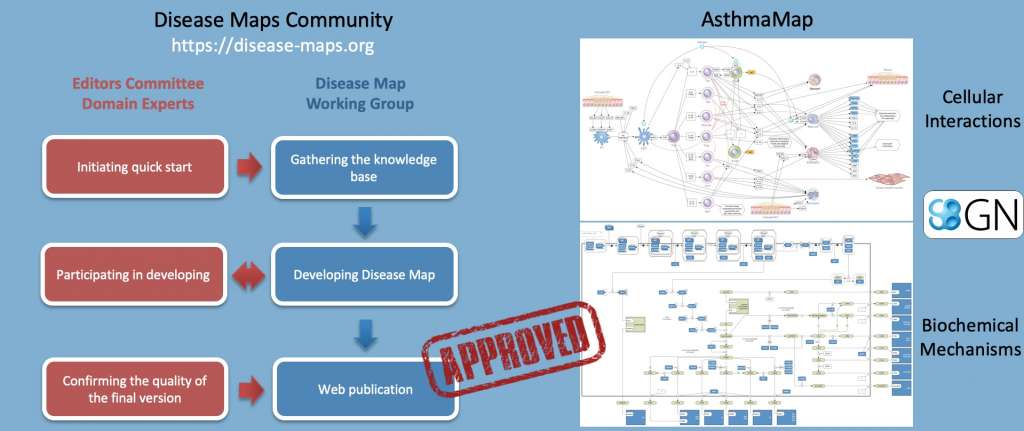 Figure 1: Development of disease maps using the Systems Biology Graphical Notation for representation of data, information, and domain expert knowledge in a human and machine readable format; example of the AsthmaMap
Figure 1: Development of disease maps using the Systems Biology Graphical Notation for representation of data, information, and domain expert knowledge in a human and machine readable format; example of the AsthmaMap
Monitoring well-being
In addition, the emergence of a wide variety of connected mobile devices has made it possible to also assess environmental and occupational exposures (e.g. nutrition, sleep, exercise, and stress) on a regular, real-time basis.14 The power of integrating these diverse types of data into a digital representation of a person has been demonstrated initially by two individuals who have managed to anticipate the occurrence of disease and take preventive measures to their benefit.15-16 An effective implementation of systems medicine has thus been made possible by scaling up the monitoring of well-being, health, and disease through the collection of billions of data points for increasing numbers of individuals who are healthy, at risk of developing disease, or in the course of disease development, with their active participation through social networks (figure 2).17-18
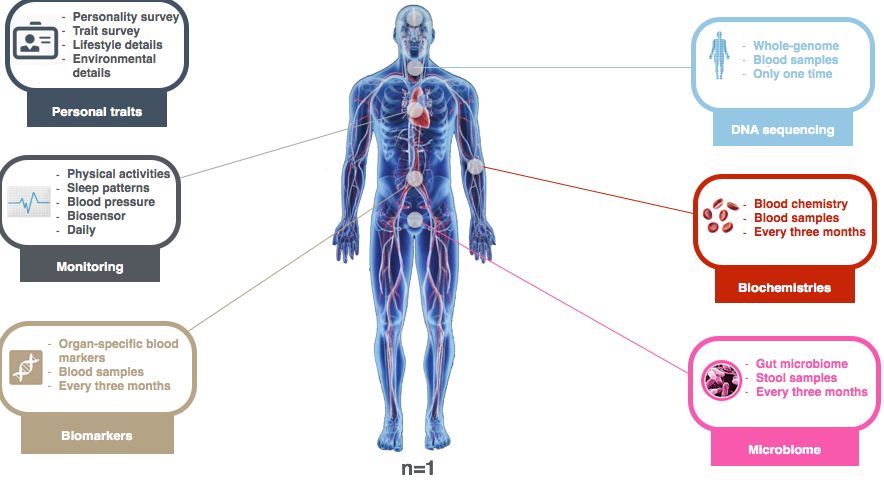 Figure 2: Monitoring well-being, health, and disease: collection of diverse data types from single individuals
Figure 2: Monitoring well-being, health, and disease: collection of diverse data types from single individuals
By monitoring individuals over a long period of time, it becomes possible to provide them with actionable recommendations to maintain their state of health and well-being, detect early events indicative of a risk or a transition to disease, and enable improvement of their personalised management, treatment and cure (figure 3).19 The expectation is that expanding the monitoring from one to millions to billions of individuals over the next generation will trigger a reversal of the escalating costs of healthcare management, drug, and diagnostic development, providing the basis for a more cost-efficient and sustainable integrated health care system.20
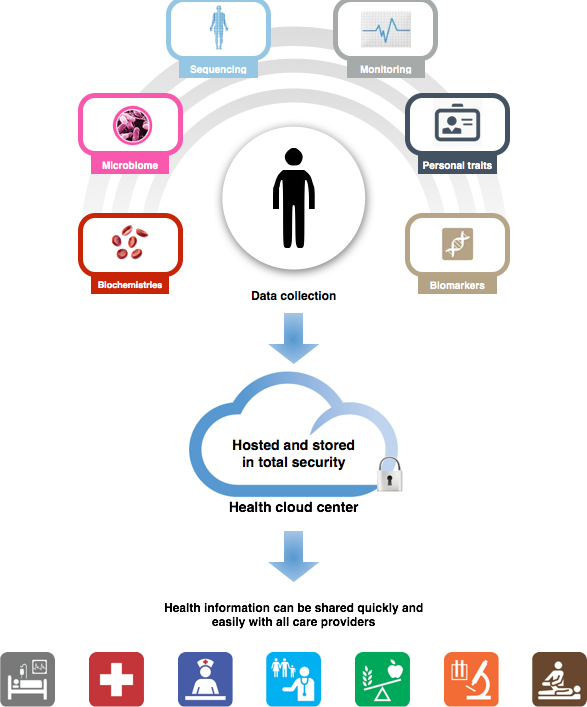
Figure 3: Monitoring well-being, health, and disease: integrated analyses to construct a digital representation of each person and deliver actionable recommendations to individuals and their care providers
Data security and training
In this context, ensuring data security and compliance with personal data and privacy protection regulations, such as the European Union General Data Protection Regulation, is important. An active participation of all stakeholders, including researchers and clinicians in academy and industry, regulatory and funding bodies, individuals and patient organisations, and the multidisciplinary training of a new generation of scientists and medical doctors, has been recognised as essential components of this ongoing transformation.11,21-22
PIONEER partnership
PIONEER will leverage these big data and systems medicine technologies. PIONEER (Prostate Cancer DIagnOsis and TreatmeNt Enhancement through the power of big data in EuRope) is a public-private partnership of 34 academic, industrial, legal, medical, and patient organisations from 9 countries across Europe, part of the Big Data for Better Outcomes Programme of the Innovative Medicines Initiative. Since May 2018, it has been supported by the European Union and the European Federation of Pharmaceutical Industries and Associations, a support which it will receive for five years total.23 PIONEER has already identified critical evidence gaps in prostate cancer care through a detailed prioritisation exercise that involved all relevant stakeholders. The consortium is set up to leverage advanced big data management and systems medicine technologies within a shared computational platform. Its mission is to provide the high-quality evidence required to support improvement in prostate cancer management and care. This will be achieved through the standardisation and integration of existing sources of data from prostate cancer patients across different stages of the disease, in close collaboration with worldwide data providers. PIONEER thus aims to contribute to overcome the current underuse of effective treatments and overuse of ineffective treatments and to enhance the quality of health and social care delivered to each individual and all prostate cancer patients and their families, while serving as an exemplar project for the dissemination of the practice of systems medicine.
The reference list can be made available to interested readers upon request by sending an email to: communications@uroweb.org.

